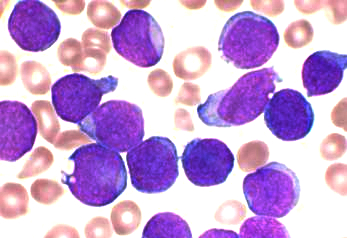
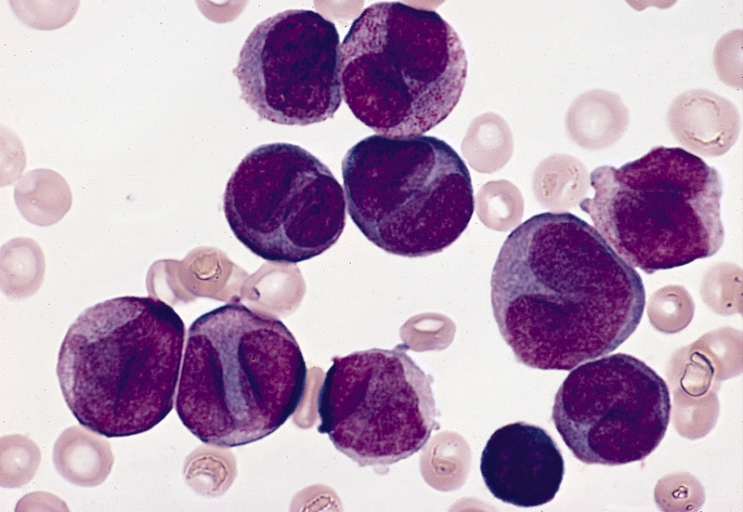
Harvard: Researchers at Harvard Medical School and Boston Children’s Hospital
were able, for the first time, to use patients’ own cells to create
cells similar to those in bone marrow and then use them to identify
potential treatments for a blood disorder. The work was published Feb. 8 in Science Translational Medicine. The team derived the so-called blood progenitor cells from two
patients with Diamond-Blackfan anemia (DBA), a rare, severe blood
disorder in which the bone marrow cannot make enough oxygen-carrying red
blood cells.
Only good, independent and reliable information about health from experts.
Showing posts with label hematology. Show all posts
Showing posts with label hematology. Show all posts
Wednesday, February 15, 2017
Wednesday, February 11, 2015
Study shows iron supplementation after blood donation shortens hemoglobin recovery time
NIH. US: A National Institutes of Health-funded study comparing low dose iron
supplementation to no supplementation in blood donors found that
supplementation significantly reduced the time to recovery of
post-donation lost iron and hemoglobin — an iron-rich protein that
carries oxygen in red blood cells throughout the body.
Thursday, January 29, 2015
FDA expands approved use of Imbruvica for rare form of non-Hodgkin lymphoma
The U.S. Food and Drug Administration today expanded the approved use
of Imbruvica (ibrutinib) for patients with Waldenström’s
macroglobulinemia (WM), a rare form of cancer that begins in the body’s
immune system. The drug received a breakthrough therapy designation for
this use.
Friday, December 26, 2014
Blood cancers could come from early mutations in life
Cancers arise from multiple acquired mutations, which presumably occur over many years. Early stages in cancer development might be present years before cancers become clinically apparent, according to a new study published in the New England Journal of Medicine.
Data where analysed from 12,380 persons blood cells DNA. Some types of mutations (permanent changes of the genes) were observed in 10% of persons older than 65 years of age but in only 1% of those younger than 50 years of age. Approximately 42% of hematologic cancers in this cohort arose in persons who had that mutations at the time of DNA sampling, more than 6 months before a first diagnosis of cancer. Analysis of bone marrow–biopsy specimens obtained from two patients at the time of diagnosis of acute myeloid leukemia revealed that their cancers arose from the earlier clones. A subset of the genes that are mutated in patients with myeloid (bone marrow) cancers is frequently mutated in apparently healthy persons; these mutations may represent characteristic early events in the development of hematologic cancers.
Data where analysed from 12,380 persons blood cells DNA. Some types of mutations (permanent changes of the genes) were observed in 10% of persons older than 65 years of age but in only 1% of those younger than 50 years of age. Approximately 42% of hematologic cancers in this cohort arose in persons who had that mutations at the time of DNA sampling, more than 6 months before a first diagnosis of cancer. Analysis of bone marrow–biopsy specimens obtained from two patients at the time of diagnosis of acute myeloid leukemia revealed that their cancers arose from the earlier clones. A subset of the genes that are mutated in patients with myeloid (bone marrow) cancers is frequently mutated in apparently healthy persons; these mutations may represent characteristic early events in the development of hematologic cancers.
Tuesday, December 23, 2014
US. FDA approves Blincyto to treat a rare form of acute lymphoblastic leukemia
The U.S. Food and Drug Administration today approved Blincyto (blinatumomab) to treat patients with Philadelphia chromosome-negative precursor B-cell acute lymphoblastic leukemia (B-cell ALL), an uncommon form of ALL.
Precursor B-cell ALL is a rapidly growing type of cancer in which the bone marrow makes too many B-cell lymphoblasts, an immature type of white blood cell. The Philadelphia chromosome is an abnormality that sometimes occurs in the bone marrow cells of leukemia patients. The National Cancer Institute estimates that 6,020 Americans will be diagnosed with ALL and 1,440 will die from the disease in 2014.
Blincyto is an example of immunotherapy, a treatment that uses certain parts of a person’s immune system to fight diseases such as cancer. Blincyto is the first approved drug that engages the body’s T-cells, a type of white blood cell or lymphocyte, to destroy leukemia cells. The drug acts as a connector between a protein called CD19, which is found on the surface of most B-cell lymphoblasts, and CD3, a protein on T-cell lymphocytes. It is intended for patients whose cancer returned after treatment (relapsed) or did not respond to previous treatment (refractory).
“Immunotherapies, especially Blincyto with its unique mechanism of action, are particularly promising for patients with leukemia,” said Richard Pazdur, M.D., director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Recognizing the potential of this novel therapy, the FDA worked proactively with the sponsor under our breakthrough therapy designation program to facilitate the approval of this novel agent.”
The FDA granted Blincyto breakthrough therapy designation, priority review and orphan product designation because the sponsor demonstrated through preliminary clinical evidence that the drug may offer a substantial improvement over available therapies; the drug had the potential, at the time the application was submitted, to be a significant improvement in safety or effectiveness in the treatment of a serious condition; and the drug is intended to treat a rare disease, respectively. Blincyto is being approved more than five months ahead of the prescription drug user fee goal date of May 19, 2015, the date the agency was scheduled to complete review of the application.
The safety and effectiveness of Blincyto were evaluated in a clinical study involving 185 adults with Philadelphia chromosome-negative relapsed or refractory precursor B-cell ALL. All participants were treated with Blincyto for at least four weeks via infusion, a method used to inject treatment into the bloodstream using a needle. Results showed 32 percent of participants had no evidence of disease (complete remission) for approximately 6.7 months.
Blincyto is being approved under the FDA’s accelerated approval program, which allows approval of a drug to treat a serious or life-threatening disease based on clinical data showing the drug has an effect on a surrogate endpoint reasonably likely to predict clinical benefit to patients. This program provides earlier patient access to promising new drugs while the company conducts confirmatory clinical trials. The FDA is requiring Blincyto’s manufacturer to conduct a study to verify that the drug improves survival in participants with relapsed or refractory Philadelphia-negative precursor B-cell ALL.
Blincyto carries a boxed warning alerting patients and health care professionals that some clinical trial participants had problems with low blood pressure and difficulty breathing (cytokine release syndrome) at the start of the first treatment, experienced a short period of difficulty with thinking (encephalopathy) or other side effects in the nervous system. The most common side effects seen in Blincyto-treated participants were fever (pyrexia), headache, swelling of tissues (peripheral edema), fever with a low number of white blood cells (febrile neutropenia), nausea, low potassium (hypokalaemia), fatigue, constipation, diarrhea and tremor.
The FDA approved Blincyto with a Risk Evaluation and Mitigation Strategy (REMS), which consists of a communication plan to inform health care providers about the serious risks and the potential for preparation and administration errors.
Blincyto is marketed by Thousand Oaks, California-based Amgen Inc.
The FDA, an agency within the U.S. Department of Health and Human Services, promotes and protects the public health by, among other things, assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices. The agency also is responsible for the safety and security of our nation’s food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products.
More with FDA
Precursor B-cell ALL is a rapidly growing type of cancer in which the bone marrow makes too many B-cell lymphoblasts, an immature type of white blood cell. The Philadelphia chromosome is an abnormality that sometimes occurs in the bone marrow cells of leukemia patients. The National Cancer Institute estimates that 6,020 Americans will be diagnosed with ALL and 1,440 will die from the disease in 2014.
Blincyto is an example of immunotherapy, a treatment that uses certain parts of a person’s immune system to fight diseases such as cancer. Blincyto is the first approved drug that engages the body’s T-cells, a type of white blood cell or lymphocyte, to destroy leukemia cells. The drug acts as a connector between a protein called CD19, which is found on the surface of most B-cell lymphoblasts, and CD3, a protein on T-cell lymphocytes. It is intended for patients whose cancer returned after treatment (relapsed) or did not respond to previous treatment (refractory).
“Immunotherapies, especially Blincyto with its unique mechanism of action, are particularly promising for patients with leukemia,” said Richard Pazdur, M.D., director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Recognizing the potential of this novel therapy, the FDA worked proactively with the sponsor under our breakthrough therapy designation program to facilitate the approval of this novel agent.”
The FDA granted Blincyto breakthrough therapy designation, priority review and orphan product designation because the sponsor demonstrated through preliminary clinical evidence that the drug may offer a substantial improvement over available therapies; the drug had the potential, at the time the application was submitted, to be a significant improvement in safety or effectiveness in the treatment of a serious condition; and the drug is intended to treat a rare disease, respectively. Blincyto is being approved more than five months ahead of the prescription drug user fee goal date of May 19, 2015, the date the agency was scheduled to complete review of the application.
The safety and effectiveness of Blincyto were evaluated in a clinical study involving 185 adults with Philadelphia chromosome-negative relapsed or refractory precursor B-cell ALL. All participants were treated with Blincyto for at least four weeks via infusion, a method used to inject treatment into the bloodstream using a needle. Results showed 32 percent of participants had no evidence of disease (complete remission) for approximately 6.7 months.
Blincyto is being approved under the FDA’s accelerated approval program, which allows approval of a drug to treat a serious or life-threatening disease based on clinical data showing the drug has an effect on a surrogate endpoint reasonably likely to predict clinical benefit to patients. This program provides earlier patient access to promising new drugs while the company conducts confirmatory clinical trials. The FDA is requiring Blincyto’s manufacturer to conduct a study to verify that the drug improves survival in participants with relapsed or refractory Philadelphia-negative precursor B-cell ALL.
Blincyto carries a boxed warning alerting patients and health care professionals that some clinical trial participants had problems with low blood pressure and difficulty breathing (cytokine release syndrome) at the start of the first treatment, experienced a short period of difficulty with thinking (encephalopathy) or other side effects in the nervous system. The most common side effects seen in Blincyto-treated participants were fever (pyrexia), headache, swelling of tissues (peripheral edema), fever with a low number of white blood cells (febrile neutropenia), nausea, low potassium (hypokalaemia), fatigue, constipation, diarrhea and tremor.
The FDA approved Blincyto with a Risk Evaluation and Mitigation Strategy (REMS), which consists of a communication plan to inform health care providers about the serious risks and the potential for preparation and administration errors.
Blincyto is marketed by Thousand Oaks, California-based Amgen Inc.
The FDA, an agency within the U.S. Department of Health and Human Services, promotes and protects the public health by, among other things, assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices. The agency also is responsible for the safety and security of our nation’s food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products.
More with FDA
Monday, October 6, 2014
Hydroxyurea
 What is sickle cell syndrome?
What is sickle cell syndrome?
Sickle cell syndrome is a group of inherited diseases caused by a mutation in a blood protein called haemoglobin. The mutation leads to changes in the shape and behaviour of red blood cells. They become hard, sticky and shaped in the form of a sickle (the sharp hooks used to cut the wheat). This causes small blood clots that block blood flow, and cause repeated painful episodes, called crisis. In the long run, this causes severe damage to the organs in the body. The clinical picture of patients with sickle cell syndrome can be remarkably different. Some patients remain virtually without complaints, while others suffer repeated crises requiring admission to hospital from early childhood. The prototype disease in the sickle cell syndrome group, is the sickle cell anaemia, which is at the same time the most common and severe in the group.
Saturday, September 27, 2014
Myeloproliferative disorders
Myeloproliferative disorders is the name for a group of conditions that cause blood cells -- platelets, white blood cells, and red blood cells -- to grow abnormally in the bone marrow.
- Polycythemia vera.
- Essential thrombocytosis
- Primary or idiopathic myelofibrosis, also known as myelosclerosis
- Chronic myelogenous leukemia (CML)
Monday, September 22, 2014
Splenomegaly
Splenomegaly is an enlargment of the spleen that can have many causes including infections, liver diseases, blood diseases, cancer.
Wednesday, August 27, 2014
Chronic Myeloid Leukemia
Also called: CML, Chronic granulocytic leukemia, Chronic myelogenous leukemia
Leukemia is cancer of the white blood cells. White blood cells help your body fight infection. Your blood cells form in your bone marrow. In leukemia, the bone marrow produces abnormal white blood cells. These cells crowd out the healthy blood cells, making it hard for blood to do its work. In chronic myeloid leukemia (CML), there are too many granulocytes, a type of white blood cell.
Most people with CML have a gene mutation (change) called the Philadelphia chromosome.
Sometimes CML does not cause any symptoms. If you have symptoms, they may include:
More about Chronic Myeloid Leukemia
Leukemia is cancer of the white blood cells. White blood cells help your body fight infection. Your blood cells form in your bone marrow. In leukemia, the bone marrow produces abnormal white blood cells. These cells crowd out the healthy blood cells, making it hard for blood to do its work. In chronic myeloid leukemia (CML), there are too many granulocytes, a type of white blood cell.
Most people with CML have a gene mutation (change) called the Philadelphia chromosome.
Sometimes CML does not cause any symptoms. If you have symptoms, they may include:
- Fatigue
- Weight loss
- Night sweats
- Fever
- Pain or a feeling of fullness below the ribs on the left side
More about Chronic Myeloid Leukemia
Chronic Lymphocytic Leukemia
Also called: CLL
Leukemia is cancer of the white blood cells. White blood cells help your body fight infection. Your blood cells form in your bone marrow. In leukemia, the bone marrow produces abnormal white blood cells. These cells crowd out the healthy blood cells, making it hard for blood to do its work. In chronic lymphocytic leukemia (CLL), there are too many lymphocytes, a type of white blood cell.
CLL is the second most common type of leukemia in adults. It often occurs during or after middle age, and is rare in children.
Usually CLL does not cause any symptoms. If you have symptoms, they may include
More about Chronic Lymphocytic Leukemia
Leukemia is cancer of the white blood cells. White blood cells help your body fight infection. Your blood cells form in your bone marrow. In leukemia, the bone marrow produces abnormal white blood cells. These cells crowd out the healthy blood cells, making it hard for blood to do its work. In chronic lymphocytic leukemia (CLL), there are too many lymphocytes, a type of white blood cell.
CLL is the second most common type of leukemia in adults. It often occurs during or after middle age, and is rare in children.
Usually CLL does not cause any symptoms. If you have symptoms, they may include
- Painless swelling of the lymph nodes in the neck, underarm, stomach, or groin
- Fatigue
- Pain or a feeling of fullness below the ribs
- Fever and infection
- Weight loss
More about Chronic Lymphocytic Leukemia
Chronic Lymphocytic Leukemia
.jpg) Author: Dr Frederick Appelbaum Fred Hutchinson Cancer Research Center Seattle 2008-07-28
Author: Dr Frederick Appelbaum Fred Hutchinson Cancer Research Center Seattle 2008-07-28Chronic Lymphocytic Leukemia (CLL) is among the most common forms of leukemia, with approximately 10,200 new cases diagnosed in the United States in 2006. The incidence of CLL increases with age and the disease is uncommon in patients less than age 50.
Chronic Myeloid Leukemia
 Author: Dr Frederick Appelbaum Fred Hutchinson Cancer Research Center Seattle 2008-07-28
Author: Dr Frederick Appelbaum Fred Hutchinson Cancer Research Center Seattle 2008-07-28 Chronic myeloid (or myelogenous) leukemia developed in approximately 4500 Americans in 2006. Although it is sometimes seen in childhood, the incidence of the disease increases with age and the median age of diagnosis is around 60 years.
Acute Promyelocytic Leukemia
Author: Dr Frederick Appelbaum Fred Hutchinson Cancer Research Center Seattle 2008-07-28
Acute Promyelocytic Leukemia (APL) is a distinct subtype of AML accounting for about 8% of cases. Patients with APL tend to be younger on average than other AML patients and are more often Hispanic. At the time of diagnosis, patients virtually always present with some evidence of a coagulation disorder, with easy bruising, petechiae or overt bleeding. A unique chromosomal translocation, t(15;17), is found in virtually every case of APL. Because of its unique clinical characteristics and response to specific agents, APL is treated differently from all other forms of leukemia.
Acute Promyelocytic Leukemia (APL) is a distinct subtype of AML accounting for about 8% of cases. Patients with APL tend to be younger on average than other AML patients and are more often Hispanic. At the time of diagnosis, patients virtually always present with some evidence of a coagulation disorder, with easy bruising, petechiae or overt bleeding. A unique chromosomal translocation, t(15;17), is found in virtually every case of APL. Because of its unique clinical characteristics and response to specific agents, APL is treated differently from all other forms of leukemia.
Acute Lymphocytic Leukemia
Author: Dr Frederick Appelbaum Fred Hutchinson Cancer Research Center Seattle 2008-07-28
Acute Lymphocytic Leukemia (ALL)
ALL is less common than AML, occurring in an estimated 4000 persons in the United States in 2006. It is the most common form of leukemia in childhood, following which the incidence drops until the age of 45, when the incidence begins to increase. Like AML, ALL is rapidly fatal if untreated. With appropriate treatment, most children with ALL can be cured of their disease.Tuesday, August 26, 2014
Acute Myeloid Leukemia
Author: Dr Frederick Appelbaum Fred Hutchinson Cancer Research Center Seattle 2008-07-28
Acute Myeloid Leukemia (AML) is the most common of the four types of leukemia, with an estimated 12,000 new cases diagnosed in the United States in 2006. AML may occur at any age, but the incidence of the disease increases as individuals get older, with the average age at diagnosis being approximately 60 years. If untreated, the average survival of patients is approximately 2 months, but with appropriate treatment, many patients can now be cured of their disease.
Acute Myeloid Leukemia (AML) is the most common of the four types of leukemia, with an estimated 12,000 new cases diagnosed in the United States in 2006. AML may occur at any age, but the incidence of the disease increases as individuals get older, with the average age at diagnosis being approximately 60 years. If untreated, the average survival of patients is approximately 2 months, but with appropriate treatment, many patients can now be cured of their disease.
Acute Myeloid Leukemia
Also called: AML, ANLL, Acute myelogenous leukemia
Leukemia is cancer of the white blood cells. White blood cells help your body fight infection. Your blood cells form in your bone marrow. In leukemia, however, the bone marrow produces abnormal white blood cells. These cells crowd out the healthy blood cells, making it hard for blood to do its work. In acute myeloid leukemia (AML), there are too many of a specific type of white blood cell called a myeloblast.
AML is the most common type of acute leukemia in adults. This type of cancer usually gets worse quickly if it is not treated. Possible risk factors include smoking, previous chemotherapy treatment, and exposure to radiation.
Symptoms of AML include:
More about Acute Myeloid Leukemia
Leukemia is cancer of the white blood cells. White blood cells help your body fight infection. Your blood cells form in your bone marrow. In leukemia, however, the bone marrow produces abnormal white blood cells. These cells crowd out the healthy blood cells, making it hard for blood to do its work. In acute myeloid leukemia (AML), there are too many of a specific type of white blood cell called a myeloblast.
AML is the most common type of acute leukemia in adults. This type of cancer usually gets worse quickly if it is not treated. Possible risk factors include smoking, previous chemotherapy treatment, and exposure to radiation.
Symptoms of AML include:
- Fever
- Shortness of breath
- Easy bruising or bleeding
- Bleeding under the skin
- Weakness or feeling tired
- Weight loss or loss of appetite
More about Acute Myeloid Leukemia
Acute Lymphocytic Leukemia
Also called: ALL, Acute lymphoblastic leukemia
Leukemia is cancer of the white blood cells. White blood cells help your body fight infection. Your blood cells form in your bone marrow. In leukemia, however, the bone marrow produces abnormal white blood cells. These cells crowd out the healthy blood cells, making it hard for blood to do its work. In acute lymphocytic leukemia (ALL), also called acute lymphoblastic leukemia, there are too many of specific types of white blood cells called lymphocytes or lymphoblasts. ALL is the most common type of cancer in children.
Possible risk factors for ALL include being male, being white, previous chemotherapy treatment, exposure to radiation, and for adults, being older than 70.
Symptoms of ALL include:
More about acute lymphoblastic leukemia
Leukemia is cancer of the white blood cells. White blood cells help your body fight infection. Your blood cells form in your bone marrow. In leukemia, however, the bone marrow produces abnormal white blood cells. These cells crowd out the healthy blood cells, making it hard for blood to do its work. In acute lymphocytic leukemia (ALL), also called acute lymphoblastic leukemia, there are too many of specific types of white blood cells called lymphocytes or lymphoblasts. ALL is the most common type of cancer in children.
Possible risk factors for ALL include being male, being white, previous chemotherapy treatment, exposure to radiation, and for adults, being older than 70.
Symptoms of ALL include:
- Weakness or feeling tired
- Fever
- Easy bruising or bleeding
- Bleeding under the skin
- Shortness of breath
- Weight loss or loss of appetite
- Pain in the bones or stomach
- Pain or a feeling of fullness below the ribs
- Painless lumps in the neck, underarm, stomach, or groin
More about acute lymphoblastic leukemia
Monday, July 28, 2014
Sickle cell anemia
Sickle cell anemia ( Also called: Hemoglobin SS disease or drepanocytosis ) is a disease in which your body produces abnormally shaped red blood cells. The cells are shaped like a crescent or sickle. They don't last as long as normal, round red blood cells. This leads to anemia. The sickle cells also get stuck in blood vessels, blocking blood flow. This can cause pain and organ damage.
A genetic problem causes sickle cell anemia. People with the disease are born with two sickle cell genes, one from each parent. If you only have one sickle cell gene, it's called sickle cell trait. About 1 in 12 African Americans has sickle cell trait.
The most common symptoms are pain and problems from anemia. Anemia can make you feel tired or weak. In addition, you might have shortness of breath, dizziness, headaches, or coldness in the hands and feet.
A blood test can show if you have the trait or anemia. Most states test newborn babies as part of their newborn screening programs.
Sickle cell anemia has no widely available cure. Treatments can help relieve symptoms and lessen complications. Researchers are investigating new treatments such as blood and marrow stem cell transplants, gene therapy, and new medicines.
More about sickle cell anemia
A genetic problem causes sickle cell anemia. People with the disease are born with two sickle cell genes, one from each parent. If you only have one sickle cell gene, it's called sickle cell trait. About 1 in 12 African Americans has sickle cell trait.
The most common symptoms are pain and problems from anemia. Anemia can make you feel tired or weak. In addition, you might have shortness of breath, dizziness, headaches, or coldness in the hands and feet.
A blood test can show if you have the trait or anemia. Most states test newborn babies as part of their newborn screening programs.
Sickle cell anemia has no widely available cure. Treatments can help relieve symptoms and lessen complications. Researchers are investigating new treatments such as blood and marrow stem cell transplants, gene therapy, and new medicines.
More about sickle cell anemia
Sickle cell anemia
Sources: Pr Frédéric GALACTEROS MD. Orphanet / Genetics Home Reference
Sickle cell disease is a group of disorders that affects hemoglobin,
the molecule in red blood cells that delivers oxygen to cells throughout
the body. People with this disorder have atypical hemoglobin molecules
called hemoglobin S, which can distort red blood cells into a sickle,
or crescent, shape.
Monday, July 21, 2014
Anemia
Anemia is a condition in which the hemoglobin content of the blood is lower than normal as a result of deficiency of nutrients (iron, vitamin...), acute or chronic blood loss (bleeding, cancer, ulcers, hemorrhoids, gastritis, nonsteroidal anti-inflammatory drugs such as aspirin or ibuprofen, excessive menstruation, multiple pregnancies, parasitic infections, acute and chronic infections, chronic renal failure, congenital hemolytic diseases (sickle cell anemia...),leukemia...
More about anemia
More about anemia
Labels
anaemia,
anemia,
bleeding,
blood,
hematology,
hemorrhage
Subscribe to:
Comments (Atom)