 CNRS: Several neurodegenerative disorders are caused by
aggregates of a single protein, alpha-synuclein, in the brain. A
French-Belgian team1 including scientists from the Institut des Neurosciences Paris Saclay
(CNRS/Université Paris-Sud) and KU Leuven has identified two specific
fiber "strains" , which take the form of "ribbons" and "cylinders", and
have been shown to cause Parkinson's disease and Multiple System Atrophy
(MSA), respectively. This work is published in Nature on 10 June 2015.
CNRS: Several neurodegenerative disorders are caused by
aggregates of a single protein, alpha-synuclein, in the brain. A
French-Belgian team1 including scientists from the Institut des Neurosciences Paris Saclay
(CNRS/Université Paris-Sud) and KU Leuven has identified two specific
fiber "strains" , which take the form of "ribbons" and "cylinders", and
have been shown to cause Parkinson's disease and Multiple System Atrophy
(MSA), respectively. This work is published in Nature on 10 June 2015.
As for many other proteins, the exact function of alpha-synuclein in the nervous system remains unknown. It may play an important role in synapses. This protein also coalesces into fibrils in the brain. These fibrils constitute Lewy bodies2, which are the hallmarks of a variety of neurodegenerative diseases such as Parkinson's disease, Multiple System Atrophy or Dementia with Lewy Bodies. A team including Ronald Melki, CNRS Senior Research at the Institut des Neurosciences Paris Saclay, has been able to show how a single protein can cause different diseases that do not affect the same cerebral functions.
Alpha-synuclein structures itself when it polymerizes into fibers. This yields highly heterogeneous solutions containing many different fibrillar forms. Because scientists cannot draw conclusions from a mixture, they had to work on a 100% pure fiber in order to obtain interpretable results.
The first step therefore consisted in isolating five types of fibers, and was the subject of an initial publication in 2013 in Nature Communications3. One of the two most important forms resembled "broad ribbons, resembling linguini" while the other looked like "solid cylindrical pasta resembling spaghetti".
These two alpha-synuclein assemblies were then injected separately into the brain and blood stream of rats. The fibers proved to be toxic to neurons and recruited the endogenous alpha-synuclein of the animals, causing the formation of further aggregates. The scientists already knew that the fibers acted like prions, but they were also able to observe that these aggregates moved along the axons of neurons.
The rodents developed two different types of synucleinopathies — Parkinson's disease and Multiple System Atrophy — depending on the inoculation they had received. This clearly demonstrates that distinct diseases result from alpha-synuclein fibers that are structurally different.
This work also shows that aggregates circulating in the blood cross the blood-brain barrier. A clearer understanding of the mechanisms underlying the propagation of alpha-synuclein assemblies could lead to the development of antibodies or molecular chaperone derivatives. These molecules, which are also proteins, could serve to prevent or slow down the formation and propagation of aggregates. This also opens the way towards new and accurate diagnostic tools for living individuals, because at present Parkinson's disease can only be diagnosed with certainty after death.
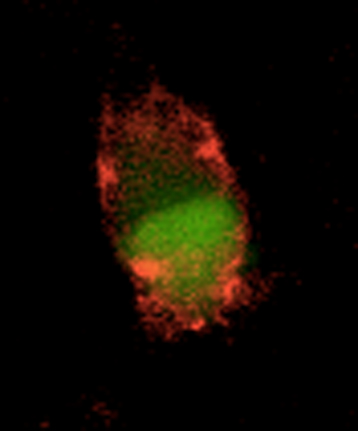
© Ronald Melki, CNRS.
Cell
in culture that has fixed alpha-synuclein fibers. The cell cytoplasm is
labeled in green, and the fixed fibers are in red. Cell observed under a
fluorescence microscope.