Northwestern: Patients with moderate to severe Crohn’s disease showed significant
improvement on a drug called ustekinumab, according to the results of
phase III clinical trials published in the New England Journal of Medicine. The study was co-authored by Stephen Hanauer, MD, the Clifford Joseph Barborka Professor of Medicine in the Division of Gastroenterology and Hepatology. Crohn’s disease is an inflammatory condition of the gastrointestinal
tract, characterized by severe abdominal pain, diarrhea and weight loss.
Current therapies, such as immunosuppressants, can help control
symptoms of the disease, but they have limited efficacy and can carry an
increased risk of infection.
Only good, independent and reliable information about health from experts.
Showing posts with label monoclonal antibody. Show all posts
Showing posts with label monoclonal antibody. Show all posts
Wednesday, January 18, 2017
Friday, January 6, 2017
Pembrolizumab in lung cancer: indication of considerable added benefit
IQWIG: Advantages notably outweigh disadvantages in comparison with docetaxel / added benefit over best supportive care not proven.Pembrolizumab (trade name: Keytruda) was initially introduced for the treatment of melanoma. Since July 2016, the monoclonal antibody has also been available for the treatment of locally advanced or metastatic non-small cell lung cancer (NSCLC) in adults whose tumours express the T-cell receptor ligand PD-L1 and who have received a prior chemotherapy regimen. The German Institute for Quality and Efficiency in Health Care (IQWiG) examined in a dossier assessment whether the drug offers an added benefit over the appropriate comparator therapy also for these patients.
Wednesday, December 14, 2016
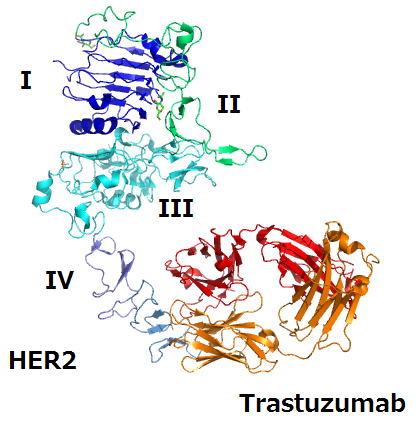
Biosimilar Drug Shows Potential as Breast Cancer Treatment
JAMA: Among women with metastatic breast cancer, treatment with a drug that
is biosimilar to the breast cancer drug trastuzumab resulted in an
equivalent overall response rate at 24 weeks compared with trastuzumab,
according to a study published online by JAMA. Biological agents, including monoclonal antibodies, have increased
the treatment options and greatly improved outcomes for a number of
cancers. However, patient access to these biologics is limited in many
countries. With impending patent expiration of some biological agents,
development of biosimilars has become a high priority for drug
developers and health authorities throughout the world to provide access
to high-quality alternatives. A biosimilar drug is a biological product
that is highly similar to a licensed biological product, with no
clinically meaningful differences in terms of safety or potency.
Wednesday, November 30, 2016
Failed Alzheimer’s trial does not kill leading theory of disease
Nature: A drug that was seen as a major test of the leading theory behind Alzheimer’s disease
has failed in a large trial of people with mild dementia. Critics of
the ‘amyloid hypothesis’, which posits that the disease is triggered by a
build-up of amyloid protein in the brain, have seized on the results as
evidence of its weakness. But the jury is still out on whether the
theory will eventually yield a treatment. Proponents
of the theory note that the particular way in which solanezumab, the
drug involved in the trial, works could have led to the failure, rather
than a flaw in the hypothesis itself. And many trials are ongoing to
test whether solanezumab — or others that target amyloid — could work in
people at risk of the disease who have not yet shown symptoms, or even
in people with Alzheimer’s, despite the latest negative result.
Thursday, February 5, 2015
Penn Medicine's New Immunotherapy Study Will Pit PD-1 Inhibitor Against Advanced Lung Cancer
Pennsylvania University. US: Penn Medicine researchers have begun a new immunotherapy trial with the “checkpoint inhibitor” known as pembrolizumab in patients with oligometastatic lung cancer—a state characterized by a few metastases in a confined area—who have completed conventional treatments and are considered free of active disease but remain at a high risk for recurrence.
Thursday, January 1, 2015
FDA approves Opdivo for advanced melanoma
The U.S. Food and Drug Administration today granted accelerated approval to Opdivo (nivolumab), a new treatment for patients with unresectable (cannot be removed by surgery) or metastatic (advanced) melanoma who no longer respond to other drugs.
Subscribe to:
Posts (Atom)