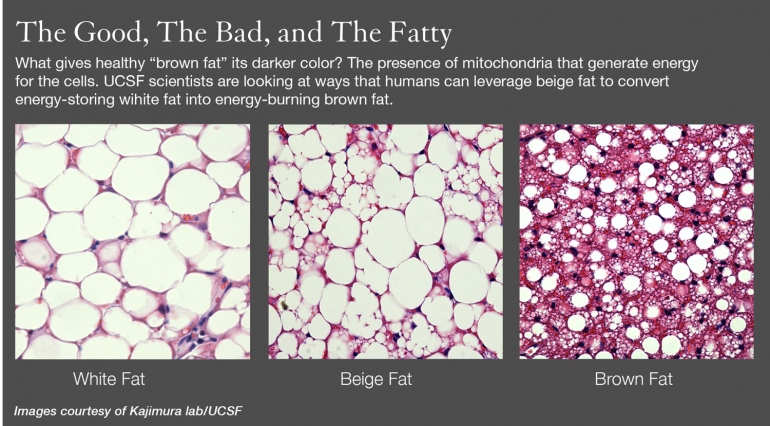
Hibernating mammals have brown fat, and human babies are born with it to help them keep warm outside the womb. Adults also have varying amounts of brown fat, but researchers weren't sure if it’s the same kind babies are born with, and if not, where it comes from.

Shingo Kajimura, PhD
A research team led by Shingo Kajimura, PhD, a UCSF assistant professor of cell and tissue biology, has for the first time isolated beige fat in adult humans and also has found new genetic markers of this beige fat.
“This is very encouraging to us; it brings us closer to engineering fat cells to fight obesity,” Kajimura said.
“We are trying to learn how to convert white fat into brown fat, and until now, it had not been demonstrated that this recruitable form of energy-burning fat is actually present in humans,” he added. Being “recruitable” means white fat can be induced to convert into fat-busting brown cells by the beige fat cells.
UCSF researchers have made significant strides in understanding the potential of brown fat. A team led by Yuriy Kirichok, PhD, discovered how a protein – called uncoupling protein 1 (UCP1) – triggers the biochemical mechanism responsible for brown fat's energy burning. And a team led by Ajay Chawla, MD, PhD, found that the immune system, rather than the nervous system, could be the key to triggering fat burning by looking at how a person shivering in cold weather (an immune response) can end up losing weight.
Now they can use these beige fat cell clones as a screening platform to identify and test different molecules that activate the development, differentiation and thermogenic (heat-producing) activity of human brown fat.
“If we have a compound that increases energy expenditure by recruiting new brown fat and activating brown fat thermogenesis, then it might work synergistically with conventional anti-obesity medications,” said Kajimura.
